COVID, Spike Protein & Nattokinase: What Does the Science Actually Say?
Introduction
Since the COVID-19 pandemic began, one question has continued to surface in both scientific research and patient discussions: what happens to the synthetic spike protein after it is introduced to the body, or the spike protein that is produced in the body after a COVID-19 infection?
Whether generated during a COVID-19 infection or through mRNA vaccination, the spike protein has become an important focus as researchers work to understand how the body processes and clears it. Some scientists have also explored whether fragments of spike protein from the virus itself might persist in certain individuals and potentially play a role in ongoing biological processes.
In his technical article “Dissolution of Spike Protein by Nattokinase,” cardiologist Dr. Peter A. McCullough reviews laboratory research examining whether the enzyme nattokinase may be capable of degrading SARS-CoV-2 spike protein under controlled conditions.
Because the original article is highly clinical and complex, this piece aims to translate that science into clearer language. We’ll explore how spike protein is produced, what researchers are learning about possible persistence following COVID infection or vaccination, and why enzymes like nattokinase have entered the conversation.
A Thoughtful Exploration Inspired by Dr. Peter A. McCullough
Based on the article discussion from:
Peter A. McCullough, MD, MPH
“Dissolution of Spike Protein by Nattokinase”
https://www.thefocalpoints.com/p/dissolution-of-spike-protein-by-nattokinase
Since the start of the pandemic, one concern has surfaced repeatedly in private conversations, clinical discussions, and independent medical commentary:
What happens to the spike protein after it’s produced in the body — and could it linger?
Dr. Peter A. McCullough addressed this question directly in his article, reviewing laboratory data examining nattokinase and its ability to degrade SARS-CoV-2 spike protein. His writing is highly clinical and technical, referencing biochemical mechanisms, enzymatic activity, and published studies in scientific journals.
For readers who are comfortable navigating medical terminology, it provides important depth. However, for many people, the language can feel dense and difficult to follow.
That is why this article was created — not to reinterpret his conclusions, but to translate the science into clearer, more understandable language.
Understanding How mRNA Vaccines Produce Spike Protein
To understand why this discussion continues, we need to revisit how mRNA vaccines were developed and deployed during the pandemic, and how synthetic spike protein functions in the body.
During the pandemic, mRNA vaccines were authorized under an Emergency Use Authorization (EUA). This pathway allows medical products to be distributed during public health emergencies once sufficient short-term safety and efficacy data are available.
However, as with any newly developed platform technology, long-term multi-year data simply did not exist at the time of widespread distribution. That does not indicate oversight — it reflects the reality that long-term biological behavior can only be observed over time as more data becomes available.
Why Vaccine mRNA Was Designed for Stability
The mRNA used in these vaccines is modified to increase stability. Normally, messenger RNA is rapidly broken down by enzymes in the body called ribonucleases. In the vaccine platform, the mRNA includes modified nucleosides that help it resist rapid degradation so that enough spike protein can be produced to trigger an immune response.
The spike protein itself was also engineered with structural modifications to stabilize it in a specific form, making it easier for the immune system to recognize.
Understanding Ongoing Health Experiences
For most individuals, the immune system clears spike protein after antibody production and cellular immunity are established. That is the expected biological outcome.
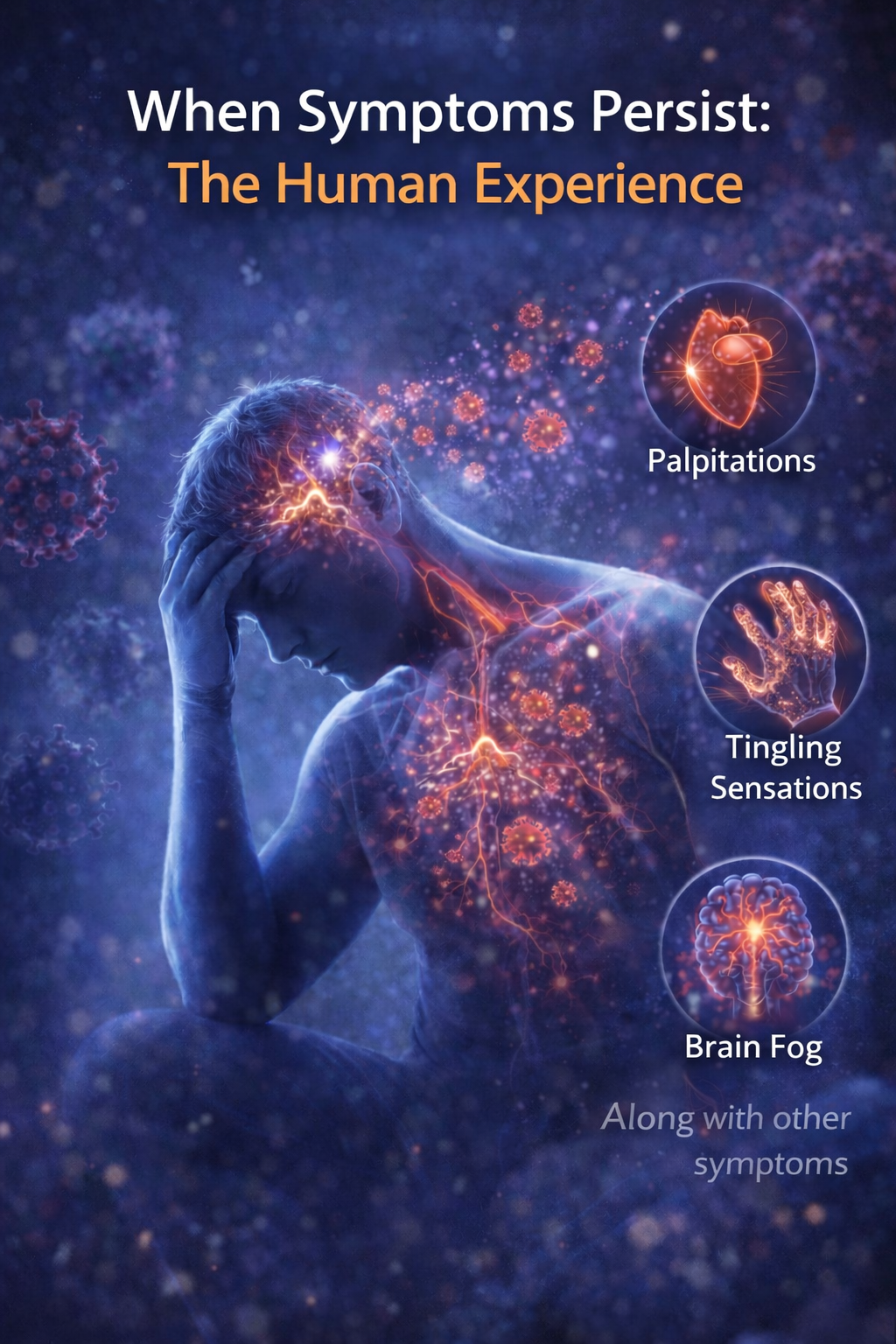
However, some individuals have reported ongoing health concerns following COVID-19 infection or vaccination. These have included fatigue, changes in exercise tolerance, chest awareness, cognitive challenges often described as “brain fog,” and other neurological or inflammatory-type experiences.
Researchers continue to study potential contributing factors and underlying mechanisms.
One area of ongoing research is whether biological factors, including protein fragments, may play a role in immune signaling in certain cases. This has not been definitively established and remains under investigation.
Enter Nattokinase: A Proteolytic Enzyme of Interest
This is where nattokinase enters the discussion.
Nattokinase is a proteolytic enzyme derived from fermented soybeans (natto). Proteolytic simply means it breaks down proteins. Historically, nattokinase has been studied for its ability to degrade fibrin — a protein involved in blood clot formation.
Because abnormal clotting patterns have been observed in both COVID-19 infection and some post-vaccination cases, nattokinase gained attention for circulatory support.
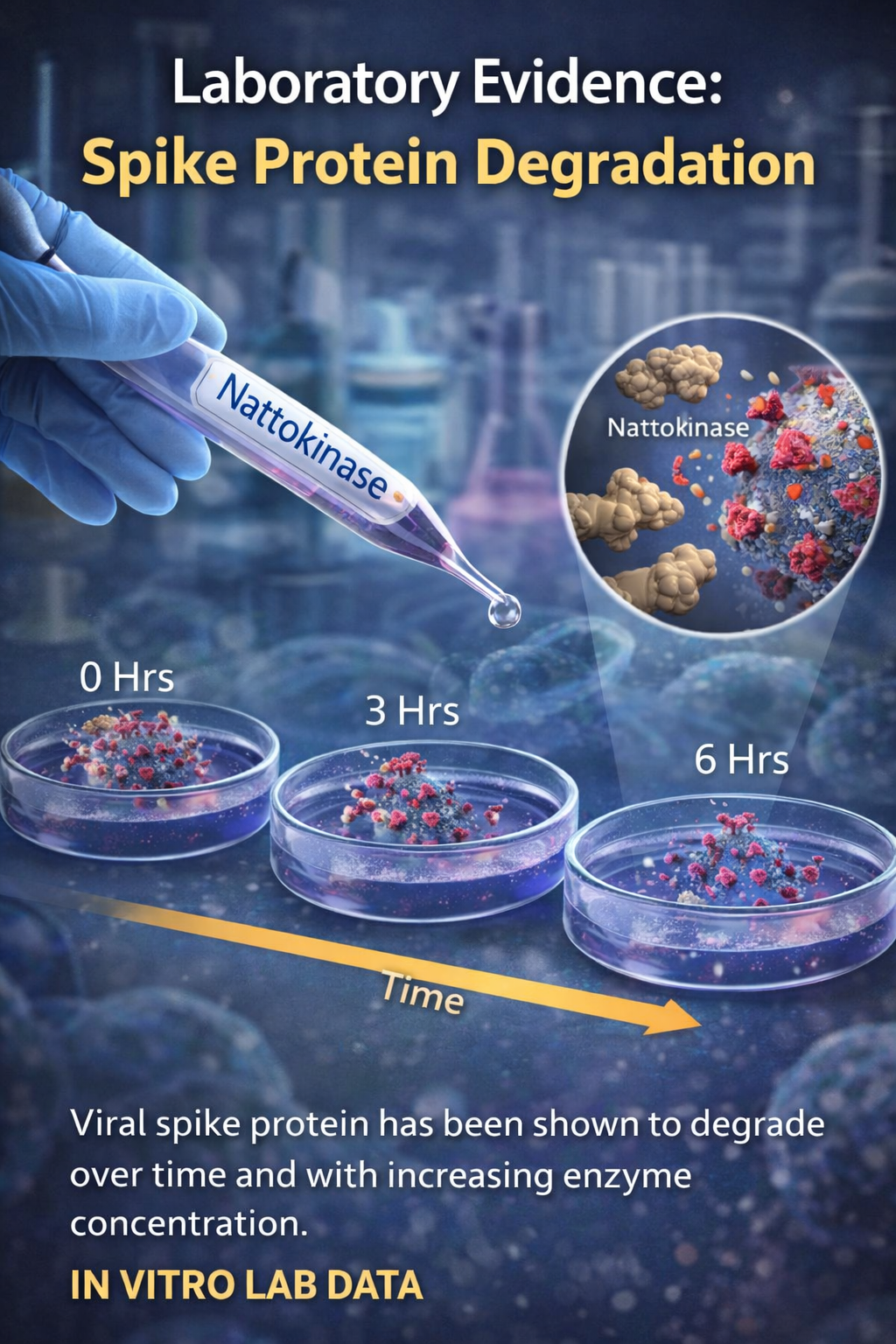
Laboratory Evidence: Spike Protein Degradation
In 2022, Tanikawa et al. published a study demonstrating that nattokinase degraded SARS-CoV-2 spike protein in vitro. “In vitro” means the experiment was conducted in a laboratory setting, not in living humans.
These findings do not prove that oral nattokinase dissolves spike protein in humans. However, they do demonstrate that spike protein is structurally vulnerable to enzymatic activity under controlled conditions.
Human Data: Systemic Effects of Nattokinase
Human studies have shown that nattokinase can influence fibrin-related markers after ingestion, suggesting systemic activity.
This has led to further discussion about how enzymes may function beyond digestion and interact with biological processes in the body.
Why Some Clinicians Consider Systemic Enzyme Therapy
Because of the laboratory findings and known biological activity of enzymes like nattokinase, some clinicians have explored their broader role in supporting normal physiological processes such as protein turnover and circulatory balance.
Given ongoing research into how the body processes proteins over time, some discussions have explored whether supporting natural metabolic pathways may be beneficial.
As with any supplement, decisions should be made in consultation with a qualified healthcare professional.
The Limits of Current Evidence
It is important to remain measured. While short-term safety and effectiveness data were available at rollout, long-term multi-year biological data continues to develop.
Ongoing pharmacovigilance — long-term safety monitoring — continues to refine understanding as more data becomes available.
The larger discussion here is not about certainty, but about inquiry.
Why Nattokinase Stands Out
Nattokinase stands out because:
- It is a protein-degrading enzyme
• It has demonstrated activity in laboratory settings
• It has documented systemic fibrinolytic activity
• It has years of supplement use data
Nattokinase stands out because it is not just another general digestive enzyme. It is a systemic proteolytic enzyme with documented fibrinolytic activity, meaning it can break down fibrin — the structural protein involved in blood clot formation. That alone makes it unique in the enzyme world.
The Role of Complementary Systemic Enzymes
Nattokinase is often discussed alongside other proteolytic enzymes such as protease, Seaprose® S, bromelain, papain, peptidase, and catalase.
These enzymes are typically explored for their roles in supporting protein metabolism, circulation, and inflammatory balance.
Science Is Still Evolving
Science rarely moves in straight lines. It unfolds step by step.
The discussion around spike protein, persistence, and enzyme-based approaches is part of that ongoing process. Over time, clearer answers may emerge.
A Practical Perspective for Individuals Seeking Answers
In the meantime, some clinicians and individuals take a practical approach. Given that systemic enzymes like nattokinase have documented biological activity, established use in circulatory support, and a generally favorable safety profile when used appropriately, many view them as an area worth further exploration.
Advocates suggest that when a strategy shows biological plausibility, laboratory support, and relatively low risk when monitored properly, it may be reasonable to explore under professional guidance.
As always, informed decision-making and consultation with qualified healthcare professionals remain essential.
Disclaimer
This article is for educational purposes only and is not intended to diagnose, treat, cure, or prevent any disease. Readers should consult a qualified healthcare professional before making decisions related to supplements or health strategies.
References
McCullough, P.A. (2023). Dissolution of Spike Protein by Nattokinase.
https://www.thefocalpoints.com/p/dissolution-of-spike-protein-by-nattokinase
Tanikawa T, Kiba Y, Yu J, et al. (2022). Degradative Effect of Nattokinase on Spike Protein of SARS-CoV-2. Molecules, 27(17):5405.
https://doi.org/10.3390/molecules27175405
Oba M, Rongduo W, Saito A, et al. (2021). Natto extract directly inhibits viral infections including SARS-CoV-2 in vitro. Biochem Biophys Res Commun, 570:21-25.
https://doi.org/10.1016/j.bbrc.2021.07.034
Kurosawa Y, Nirengi S, Homma T, et al. (2015). A single-dose of oral nattokinase potentiates thrombolysis and anti-coagulation profiles. Scientific Reports, 5:11601.
https://doi.org/10.1038/srep11601









0 Comments